Published in Cell’s Molecular Therapy – Nucleic Acids
Abstract
The use of T cells from healthy donors for allogeneic chimeric antigen receptor T (CAR-T) cell cancer therapy is attractive because healthy donor T cells can produce versatile off-the-shelf CAR-T treatments. To maximize safety and durability of allogeneic products, the endogenous T cell receptor and major histocompatibility complex class I molecules are often removed via knockout of T cell receptor beta constant (TRBC) (or T cell receptor alpha constant [TRAC]) and B2M, respectively. However, gene editing tools (e.g., CRISPR-Cas9) can display poor fidelity, which may result in dangerous off-target mutations.
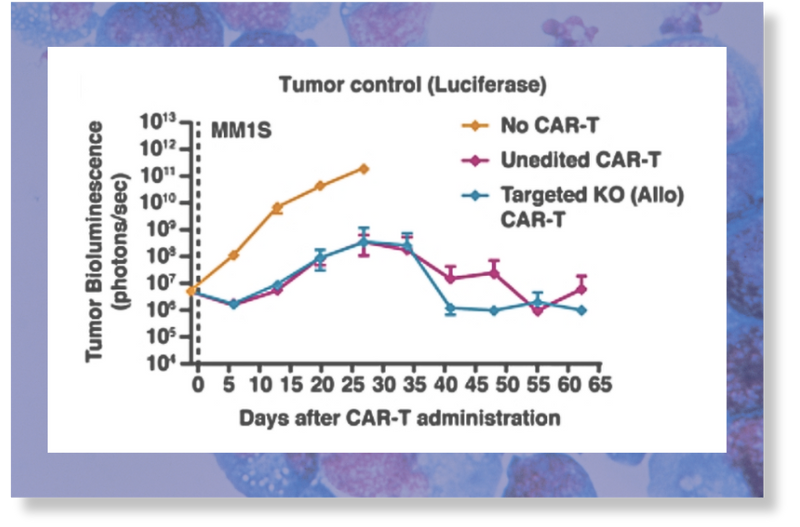
Allogeneic anti-BCMA CAR-T cells produced using Cas-CLOVER showed anti-tumor efficacy comparable with non-gene-edited CAR-T cells in the systemic MM.1S xenograft model of multiple myeloma.
Additionally, many gene editing technologies require T cell activation, resulting in a low percentage of desirable stem cell memory T cells (TSCM). We characterize an RNA-guided endonuclease, called Cas-CLOVER, consisting of the Clo051 nuclease domain fused with catalytically dead Cas9. In primary T cells from multiple donors, we find that Cas-CLOVER is a high-fidelity site-specific nuclease, with low off-target activity. Notably, Cas-CLOVER yields efficient multiplexed gene editing in resting T cells. In conjunction with the piggyBac transposon for delivery of a CAR transgene against the B cell maturation antigen (BCMA), we produce allogeneic CAR-T cells composed of high percentages of TSCM cells and possessing potent in vivo anti-tumor cytotoxicity.
Additional Resources
More to help guide your gene editing implementation journey



