Lexington, KY and Bloomington, IN: A pioneering study, authored by scientists from Demeetra and Indiana University, has been selected for the MDPI Journals Editor’s Choice Articles, highlighting the impact of advanced gene editing in synthetic biology to produce biopesticides.

Published in Fermentation in 2023, Generation of a Culex Male Mosquito Sex-Separation RNAi Yeast Strain Using Cas-CLOVER and Super PiggyBac Engineering in Saccharomyces cerevisiae, showcases the generation of a RNAi yeast strain, which may be used to accelerate adoption of a mosquito control method called sterile insect technique (SIT).
MDPI states, “Editor’s Choice articles are based on recommendations by the scientific editors of MDPI journals from around the world. Editors select a small number of articles recently published in the journal that they believe will be particularly interesting to readers, or important in the respective research area. The aim is to provide a snapshot of some of the most exciting work published in the various research areas of the journal.”
Demeetra’s Cas-CLOVER Used in combination with piggyBac Transposase
Dr. Molly Duman-Scheel, Navari Family Professor in the Department of Medical and Molecular Genetics at the Indiana University School of Medicine, initially used RNAi as a tool to study genes involved with mosquito development. With her extensive genetic knowledge, she shifted to translational research using RNAi as the insecticide itself. Due to the steep cost and limited environmental stability of RNAi, Dr. Duman-Scheel turned to expressing RNAi in yeast as a production system which overcomes both problems.
According to Duman-Scheel, “In studies led by Dr. Keshava Mysore, our group was initially able to demonstrate proof of concept with laboratory yeast strains, but high level RNA expression in a robust commercial-ready stable yeast strain suitable for industrial scale-up was required for commercialization.”
A More Precise Alternative to CRISPR/Cas9
Through a collaboration with Demeetra, which produced two publications in 2023, Dr. Duman-Scheel gained access to advanced gene editing technologies and exceptional know-how. “We wanted to engineer the ideal yeast platform for stable RNAi expression” said Dr. Corey Brizzee, Director of Gene Editing at Demeetra. After obtaining the RNAi sequences, Demeetra scientists led by Brizzee used Cas-CLOVER targeted nuclease, a more precise alternative to CRISPR/Cas9, to knock out multiple genes that could be used to select for high expressing clones.
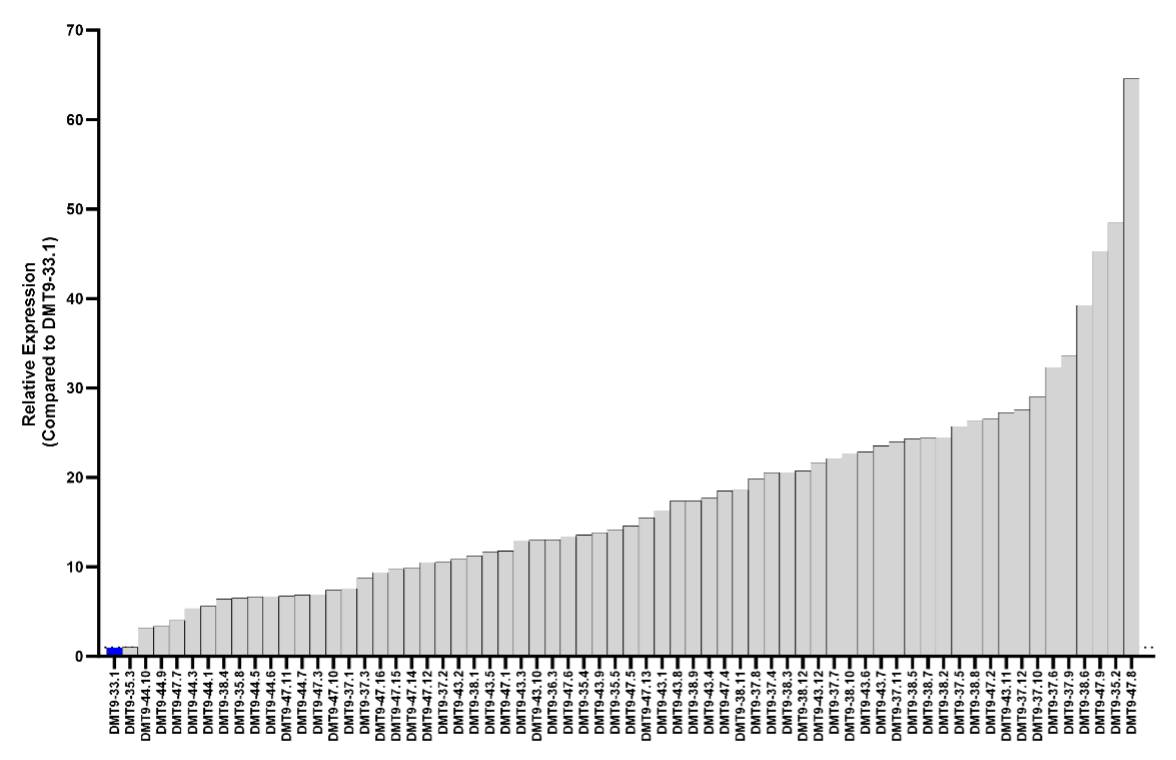
Using the edited yeast strain as a foundation, piggyBac transposase was employed to stably deliver a wide range of gene copies at different integration sites.
“piggyBac’s integration preference is non-coding regions of open chromatin, where transgenes are highly expressed. This makes the transposase perfect for bioprocessing in which we can obtain a dynamic range of expression, including far above the level of transient expression” Brizzee added.
Study Uses Sterile Insect Technique (SIT) as Biological Control
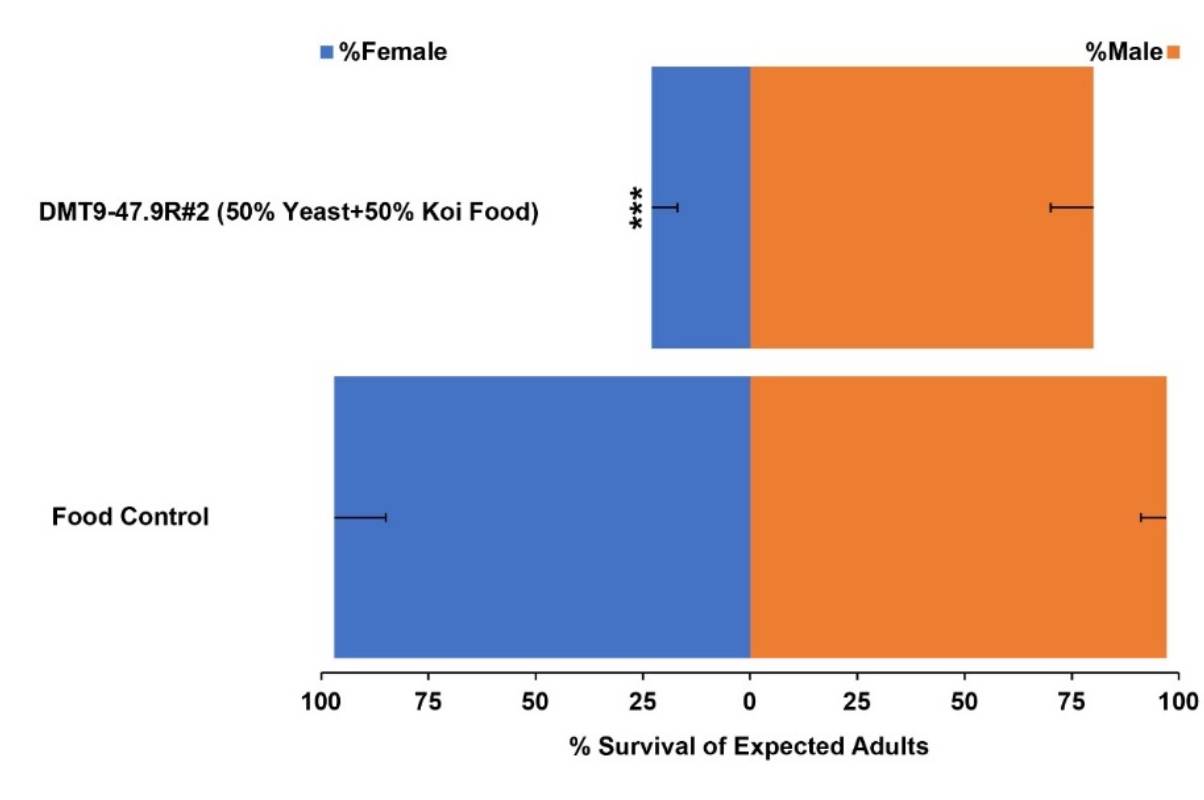
Mosquito-borne diseases claim hundreds of thousands of lives and cause billions in economic damage annually. Traditional mosquito control methods face challenges like insecticide resistance and environmental concerns. The sterile insect technique (SIT) is a form of biological control that involves the mass-rearing and sterilization of male insects, which are then released into the wild to mate with wild females. Since these couplings do not produce offspring, repeated releases of sterile males can significantly reduce or even eliminate the targeted pest population over time. One of the key inefficiencies to the process is the exhaustive sorting of male from female larvae. The stable RNAi expressing yeast strains facilitate male sex separation by selectively killing female larvae that consume the yeast.
According to Duman-Scheel, “The sex-sorting yeast will facilitate mass-production of male mosquitoes that can be used in SIT programs, a welcome relief and considerable financial savings for mosquito control programs that are currently sorting the mosquitoes by hand.”
The implications of this research extend beyond mosquito control. The Cas-CLOVER and piggyBac technologies have been established in yeast, mammalian cells and plants, providing a scalable and efficient platform for any synthetic biology application. This not only opens new avenues for disease vectors, as well as environmental and agricultural pest management but also bioprocessing of therapeutics.
About Demeetra
Based in Lexington, KY, Demeetra’s core focus is developing and optimizing gene editing technologies in commercially applicable systems. Our team of peer-review published scientific staff offers expert technical support, proprietary protocols, and extensive know-how. We transfer this knowledge to our partners and provide simple commercial licenses with freedom to operate. We invite researchers and industry partners to join us in exploring the vast potential of Cas-CLOVER nuclease and piggyBac transposase technologies.
About Indiana University
Indiana University (IU) is one of the nation’s leading public research universities, with 90,000 students across 930+ academic programs, and nine campuses . Since 1820, Indiana University has helped students create brighter futures while also driving innovation, from breakthroughs in DNA technology to cancer research to trailblazing cultural programs and resources. IU is home to world-class academics with the country’s largest medical school, the world’s first school of philanthropy, the top-ranked Kelley School of Business and O’Neill School of Public and Environmental Affairs, and the Luddy School of Informatics, Computing and Engineering, the nation’s first school of informatics. The university’s campuses are united by IU 2030, an aspirational vision for a bold and ambitious future focused on student success and opportunity, transformative research and creativity, and service to the state of Indiana and beyond. Learn more at iu.edu.
