From gene to clinical material. One partner. One path.
Eliminate Rebuilds. Eliminate Re-Qualification. Eliminate Friction.
Traditional development models fragment CLD and manufacturing across multiple vendors and technology systems. The results often include:
The Demeetra Integrated Advantage
One Continuous Development Strategy
Discovery → CLD → Upstream Development → GMP Manufacturing
Instead of transferring between disconnected systems or switching editing platforms before commercialization, our partners advance the exact same characterized cell lines into GMP production.
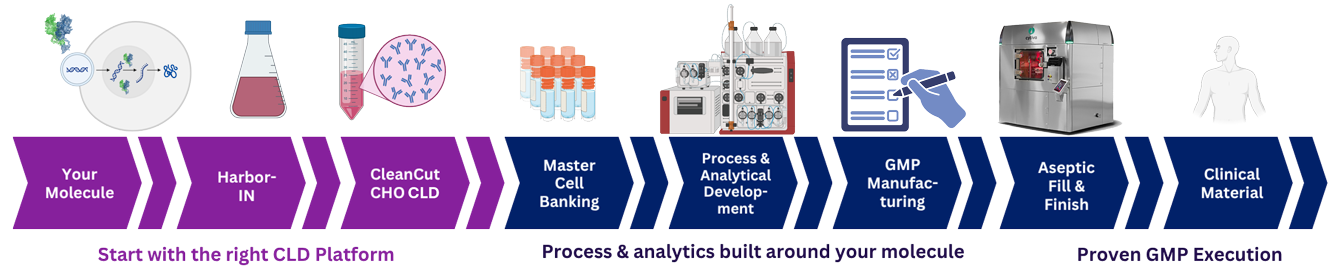
This enables:
Your molecule stays on a single, coherent development path – technically, strategically, and with clear freedom to operate.
Protecting What Matters Most
Demeetra’s integrated CLD-to-GMP solution is designed to safeguard the core drivers of program success as you advance toward clinical manufacturing:
Built for Emerging and Established Biopharma
Whether you are advancing your first biologic or expanding a multi-asset pipeline, our integrated model provides:
Start with the End in Mind
Manufacturing should not require starting over. With Demeetra’s Integrated Cell Line Development to GMP Manufacturing service, your program advances on a single, continuous technical and licensing foundation.
Let’s build your next clinical asset on an integrated path that protects both time and value.
