Increase Antibody Potency without Compromising Manufacturability.
Create higher-value, Fc-enabled antibody therapeutics while maintaining manufacturability with the CleanCut GS ADCC+ therapeutic optimization platform
CleanCut GS ADCC+ CHO Cells.
CleanCut GS ADCC+ is a next-generation CHO host cell platform designed for developers who want to improve the therapeutic performance of Fc-enabled antibodies while preserving the speed and productivity required for cell line development and scale-up.
Molecule classes and applications that can benefit from CleanCut GS ADCC+
- Monoclonal antibodies with Fc effector function
- Fc-enabled bispecific antibodies
- Oncology, inflammation, autoimmune, selected infectious
disease indications
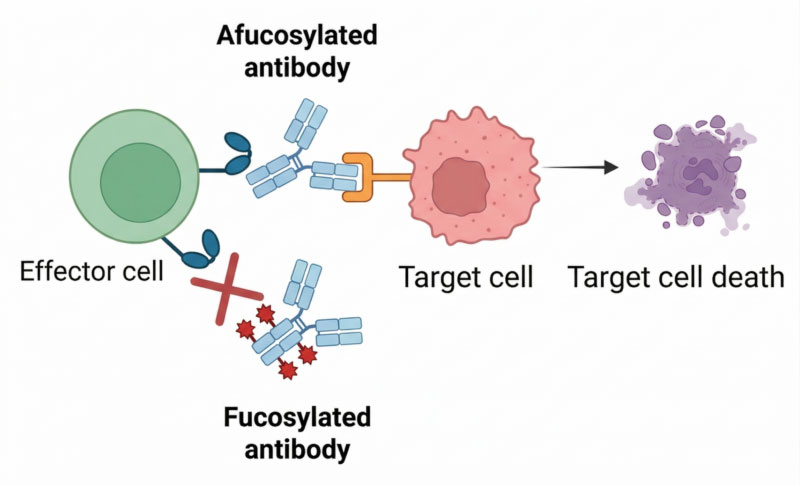
Built on Demeetra’s CleanCut GS background, this platform combines a dual GS knockout foundation with a biallelic FUT8 knockout to enable production of afucosylated antibodies with enhanced antibody-dependent cellular cytotoxicity (ADCC) potential—without the productivity and growth penalties often associated with glycoengineered hosts.
Enhanced ADCC that can increase program value
The most important outcome is functional, not just analytical.
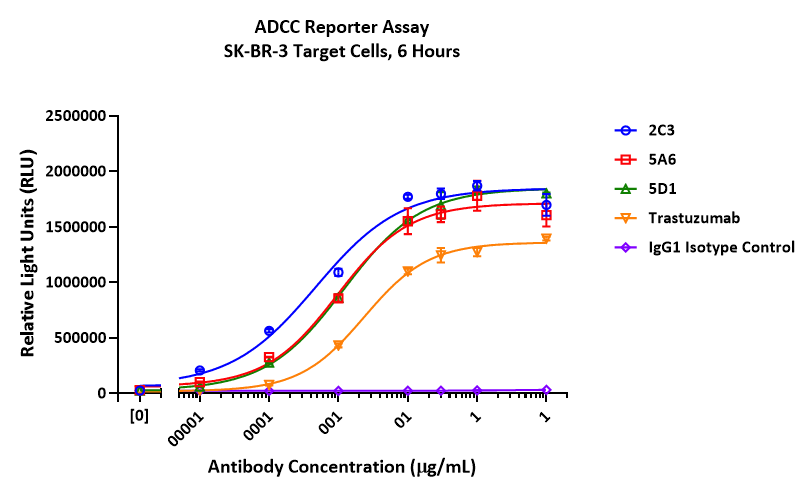
In an ADCC reporter assay, afucosylated trastuzumab produced from the top CleanCut GS ADCC+ clones (2C3, 5A6, and 5D1) showed substantially enhanced ADCC activity and potency relative to control (trastuzumab).
| Measured EC50 | |
|---|---|
| CleanCut GS control | 0.024 µg/mL |
| ADCC+ clone 2C3 | 0.005 µg/mL |
| ADCC+ clone 5A6 | 0.010 µg/mL |
| ADCC+ clone 5D1 | 0.012 µg/mL |
CleanCut GS ADCC+ maintains growth and productivity
A major differentiator of CleanCut GS ADCC+ is that the platform creates afucosylated antibodies while maintaining strong manufacturing performance.
Top clones achieved titers ranging from approximately 5.7–5.8 g/L in fed-batch spin-tube cultures, demonstrating that enhanced antibody performance can be achieved without sacrificing manufacturability.
Optimizing Your Antibody Therapeutics?
Access CleanCut GS ADCC+ as a differentiated host cell platform for internal use, transfer to your CDMO of choice, or as part of a timeline-compressed CLD-to-GMP solution with Demeetra and its manufacturing partner NorthX Biologics.
